News

Regenative Labs And AAPC Announce Collaboration To Further Healthcare Compliance
Aug 01, 2022
PENSACOLA, Fla., Aug. 1, 2022 /PRNewswire/ — Regenative Labs, a leading HCT/P manufacturer, and AAPC, the nation’s largest medical coding, training, and… Read More >
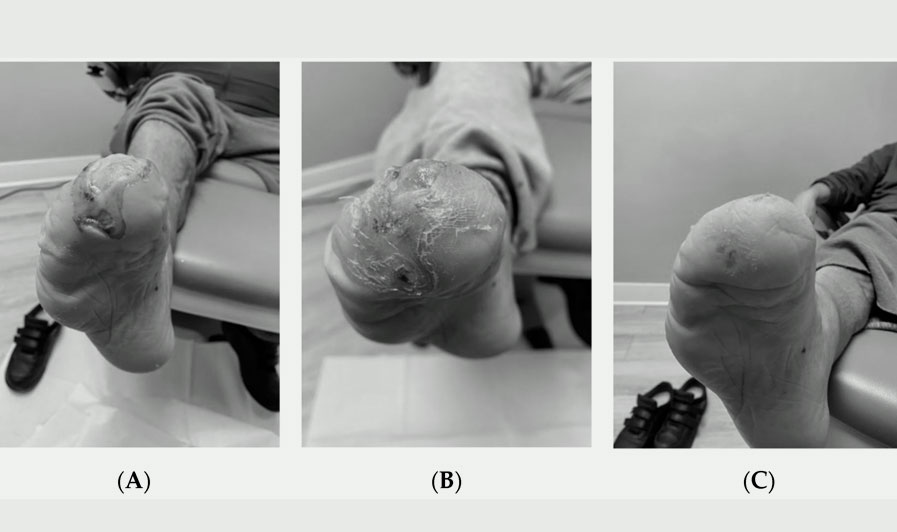
Application of Dehydrated Amniotic Membrane Allografts in Advanced Diabetic Foot Ulceration: Case Report and Review of Literature
Jul 20, 2022
by Catherine Becker 1,*, Matthew Regulski 2, Scott Martin 1 and Tyler Barrett 1 Regenative Labs, 1700 Main Street, Pensacola,… Read More >

Institutional Review Board Approves Regenative Labs’ Protocol For Key Data Collection
Jul 08, 2022
Regenative Labs CEO predicts this will have significant implications to advance multiple fields. PENSACOLA, Fla., July 8, 2022 /PRNewswire/ —… Read More >

Bone Marrow Aspirate Concentrate for the Treatment of Knee Osteoarthritis: A Systematic Review
Jul 08, 2021
Laura E Keeling 1, John W Belk 2, Matthew J Kraeutler 3, Alexandra C Kallner 4, Adam Lindsay 2, Eric C McCarty 2, William F Postma 1 PMID: 34236913 DOI: 10.1177/03635465211018837 Link to original article: Bone… Read More >

A Letter from the CEO
Jun 15, 2021
Regenative Labs to-date produces the first and only Wharton’s jelly allograft to be recognized as a 361 HCT/P by any… Read More >

Regenative Labs: Medicare Approval for Wharton’s Jelly Reimbursement
Aug 05, 2020
PENSACOLA, FLA. (PRWEB) AUGUST 05, 2020 Regenative Labs has received approval from the Centers for Medicare & Medicaid Services (CMS)… Read More >